|
3/29/2024 0 Comments Atomic theory definition 
Particle locations in quantum mechanics are not at an exact position, and they are described by a probability density function. On the atomic scale, physicists have found that quantum mechanics describes things very well on that scale. The classical description cannot be used to describe things on the atomic scale. Due to the quantum nature of electrons, the electrons are not pointing particles, and they are smeared out over the whole atom. It may seem that the space and the matter are empty, but it is not. Together, these electrons weigh only a fraction (let’s say 0.05%) of the entire atom. But this “huge” space is occupied primarily by electrons because the nucleus occupies only about 1721×10 −45 m 3 of space. Assuming a spherical shape, the uranium atom has a volume of about 26.9 ×10 −30 m 3. The Van der Waals radius, r w, of an atom is the radius of an imaginary hard-sphere representing the distance of the closest approach to another atom. For uranium atom, the Van der Waals radius is about 186 pm = 1.86 ×10 −10 m. The volume of an atom is about 15 orders of magnitude larger than the volume of a nucleus. Protons and neutrons are most likely found in the same space, at the central point. Furthermore, the nucleus is responsible for most of the mass of the atom.Ī figurative depiction of the helium-4 atom with the electron cloud in shades of gray. He postulated that the positive charge in an atom is concentrated in a small region (in comparison to the rest of the atom) called a nucleus at the center of the atom with electrons existing in orbits around it. In other words, the nucleus occupies only about 10 -12 of the total volume of the atom or less (the nuclear atom is largely empty space), but it contains all the positive charge and at least 99.95% of the total mass of the atom.īased on these results, Ernest Rutherford proposed a new model of the atom. In Rutherford’s atom, the diameter of its sphere (about 10 -10 m) of influence is determined by its electrons. Its positive charge and most of its mass is concentrated in a small region called an atomic nucleus. Scientists discovered that every atom contains a nucleus (whose diameter is of the order of 10 -14m.). Geiger-Marsden experiments were a landmark series of experiments. Previous experiments showed that the deflections had to be electrical or perhaps magnetic in origin.Īs a result, Rutherford abandoned the Thomson model. This force could be provided as an only result of a collision with a massive target or from an interaction with an electric or magnetic field of great strength. Rutherford assumed that to deflect the alpha particle backward, and there must be a very large force. “It is almost as incredible as if you had fired a fifteen-inch shell at a sheet of tissue paper, and it came back and hit you.” 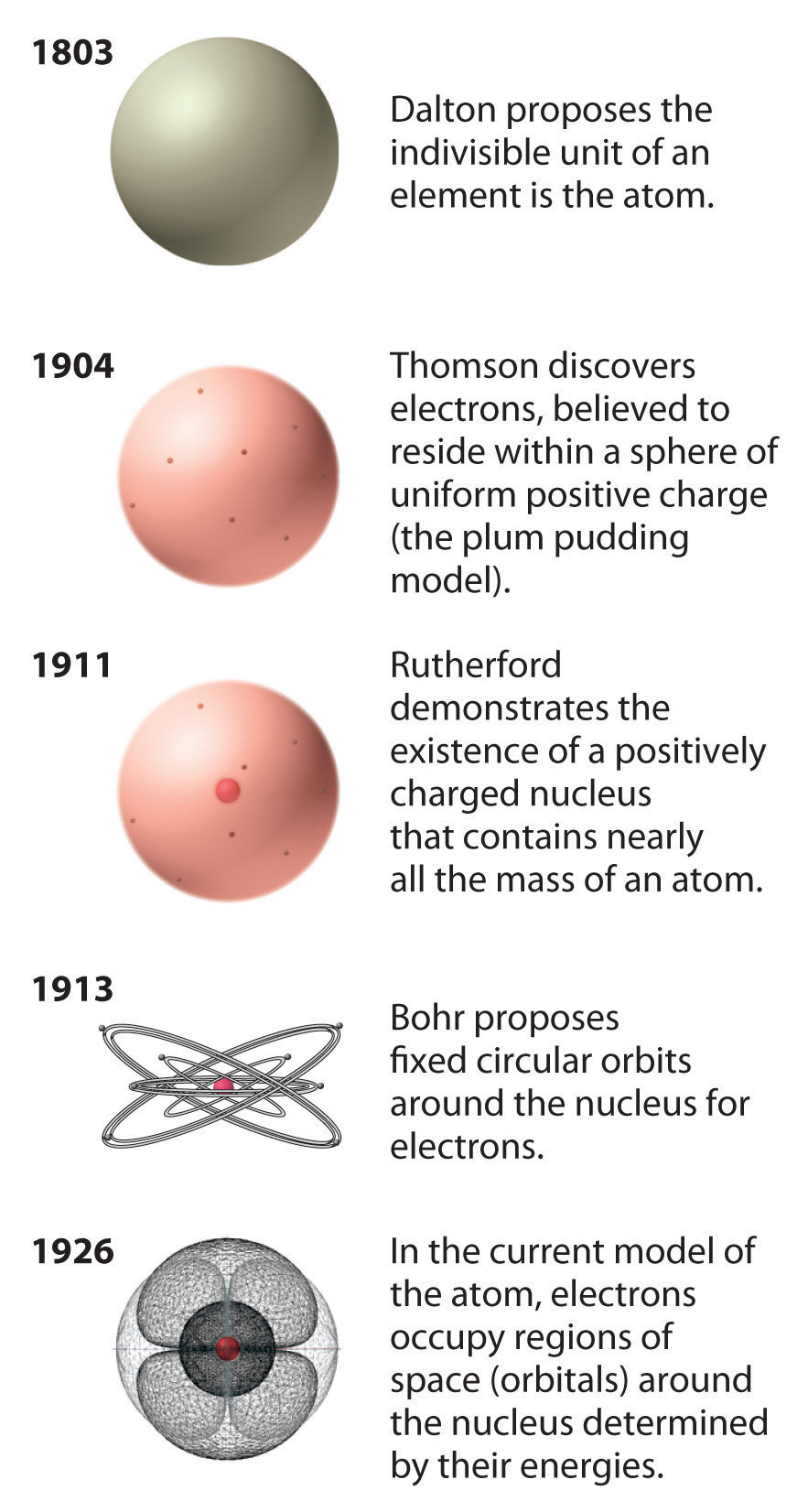 
A small fraction of them is scattered through very large angles, approaching 180° (i.e., they recoiled backward). But in the experiment, Geiger and Marsden saw that most of the particles were scattered through rather small angles, but this was a big surprise. Because of their relatively much greater mass, alpha particles are not significantly deflected from their paths by the electrons in the metal’s atoms.Īccording to Thomson’s model, if an alpha particle were to collide with a plum-pudding atom, it would just fly straight through, its path deflected by at most a fraction of a degree. Alpha particles, which are about 7300 times more massive than electrons, have a positive charge of +2e. Alpha particles are energetic nuclei of helium (usually about 6 MeV). A narrow collimated beam of alpha particles was aimed at a gold foil of approximately 1 μm thickness (about 10,000 atoms thick). Rutherford’s idea was to direct energetic alpha particles at a thin metal foil and measure how an alpha particle beam is scattered when it strikes a thin metal foil. The Geiger–Marsden experiments were performed between 19 by Hans Geiger (of Geiger counter fame) and Ernest Marsden (a 20-year-old student who had not yet earned his bachelor’s degree) under the direction of Ernest Rutherford. Rutherford’s new model for the atom is based on the experimental results obtained from the Geiger-Marsden experiments (also called the Rutherford gold foil experiment). The Rutherford model of the atom is a model of the atom devised by the British physicist Ernest Rutherford. 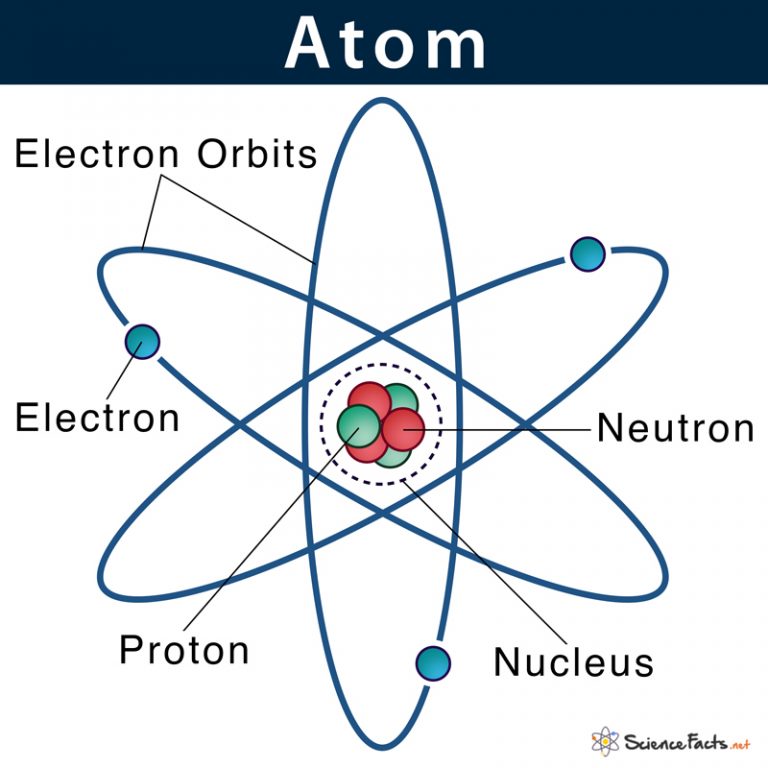
Furthermore, the nucleus is responsible for most of the mass of the atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
